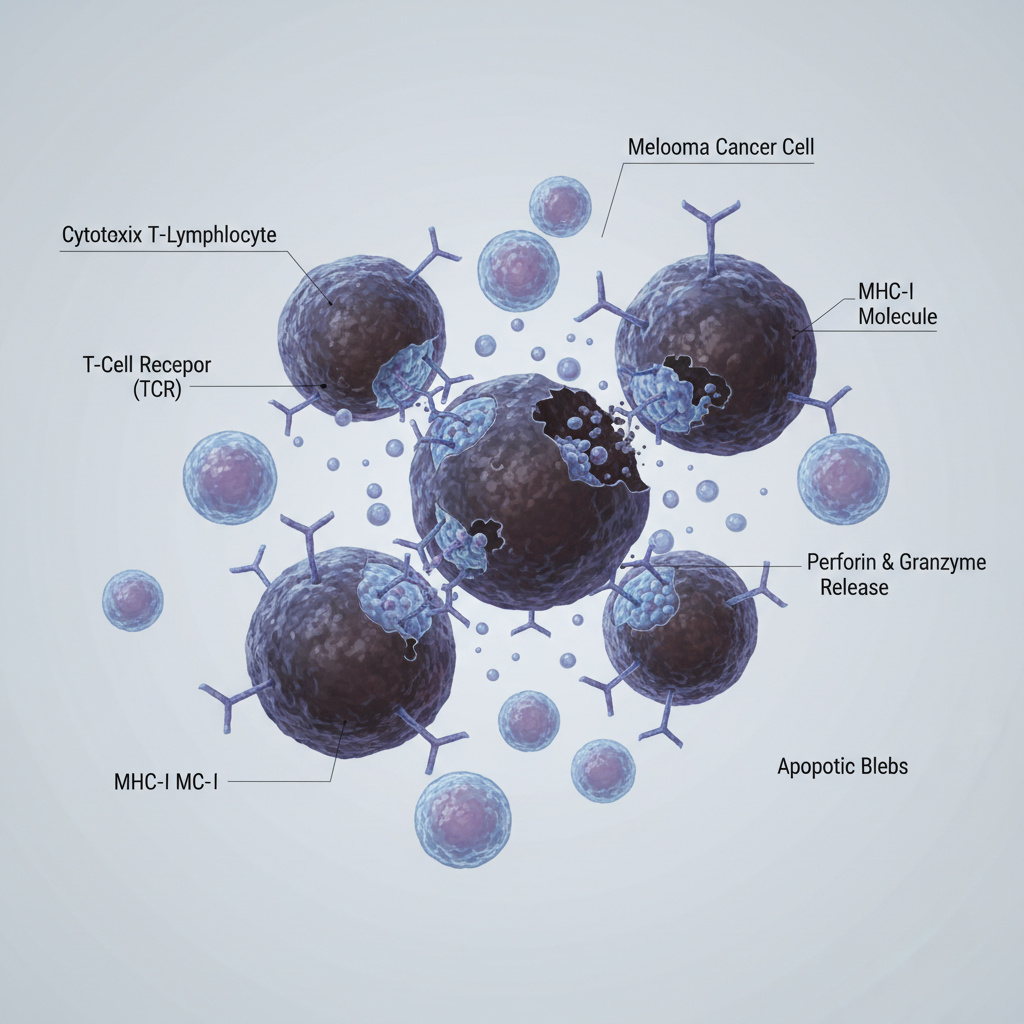
CAR-T cell therapy is one of the most advanced treatment options for certain blood cancers. This innovative approach uses the patient’s own immune system to identify and destroy cancer cells.
Unlike chemotherapy or radiation therapy, CAR-T treatment uses genetically modified T-cells that are specifically engineered to recognize cancer cells and activate an immune response against them.
One of the first CAR-T therapies approved for clinical use was Kymriah (tisagenlecleucel), developed by Novartis and approved in 2017. Since then, CAR-T therapy has become an important treatment option for patients with relapsed or refractory blood cancers when standard treatments are no longer effective.
CAR-T therapy is considered a personalized treatment. Each therapy is manufactured individually using the patient’s own immune cells. For this reason, CAR-T therapy requires specialized medical centers and a multidisciplinary team of experienced specialists.
Conditions Treated with CAR-T Therapy
The primary use of CAR-T therapy is in hematologic oncology. However, in recent years, the scope of CAR-T therapy has expanded to include additional diseases and emerging clinical applications.
Hematologic Oncology (Primary Indication)
CAR-T therapy is most commonly used for patients with relapsed or refractory blood cancers.
Lymphomas
CAR-T therapy for lymphoma is commonly used in patients with:
- Diffuse Large B-Cell Lymphoma (DLBCL)
- Primary Mediastinal B-Cell Lymphoma
- Relapsed or refractory lymphomas
These conditions often require advanced treatment options after multiple lines of therapy.
Mantle Cell Lymphoma
CAR-T therapy is also used in patients with mantle cell lymphoma who have undergone several prior treatments. Therapies such as Tecartus (brexucabtagene autoleucel) have shown promising results in these patients.
Acute Lymphoblastic Leukemia (ALL)
CAR-T therapy for leukemia is used in children, adolescents, and young adults with relapsed or refractory acute lymphoblastic leukemia. In some cases, this therapy may be considered an alternative to bone marrow transplantation.
Multiple Myeloma
CAR-T therapy for multiple myeloma has developed rapidly in recent years. BCMA-targeted CAR-T therapies have demonstrated high response rates in patients with relapsed disease, particularly after multiple lines of treatment.
Autoimmune Diseases (Experimental Direction)
In addition to cancer treatment, CAR-T therapy is currently being studied for severe autoimmune diseases.
Emerging research includes:
- Systemic lupus erythematosus
- Systemic sclerosis
- Rheumatoid arthritis
In these cases, CAR-T cells target abnormal B-cells responsible for producing harmful antibodies. This approach is currently considered experimental and is mainly available in clinical trial settings.
Physicians Specializing in CAR-T Therapy
CAR-T therapy is one of the most complex treatments in hematologic oncology. It requires specialists with experience in:
- Hematologic malignancies
- Bone marrow transplantation
- Cellular therapy
- Immunotherapy
Our CAR-T program includes physicians experienced in treating:
- Relapsed lymphoma
- Acute leukemia
- Multiple myeloma
- Complex cases after bone marrow transplantation
Specialists involved in CAR-T therapy include:
- Prof. Yair Herishanu — Hematologic Oncology, CAR-T Therapy
- Prof. Irit Avivi — Bone Marrow Transplantation, Cellular Therapy
- Prof. Ronit El-Hasid — Pediatric Hematology Oncology, CAR-T
- Prof. Ram Rom — Hematologic Oncology, Cellular Therapy
- Prof. Arnon Nagler — Bone Marrow Transplantation
- Dr. Avigdor Avraham — Hematologic Oncology
- Dr. Dror Levin — Cellular Therapy
- Dr. Freddy Aviv — Hematologic Oncology
- Dr. Hava Perry — Hematologic Oncology
The multidisciplinary team evaluates patients and guides them through each stage of treatment.




















Why Patients Choose CAR-T Therapy in Israel
CAR-T therapy is a highly specialized treatment available in a limited number of medical centers worldwide. Israel has become a destination for patients seeking advanced cellular therapies.
Patients consider CAR-T therapy in Israel for several reasons:
- Access to advanced CAR-T technologies
- Experienced specialists in complex cases
- Personalized treatment planning
- Multidisciplinary medical teams
- Support for international patients
Each patient undergoes an individual evaluation before treatment decisions are made.
Who May Be Eligible for CAR-T Therapy
CAR-T therapy may be considered in the following situations:
- Lack of response to standard treatment
- Relapsed disease
- Limited treatment options
- Ineligibility for bone marrow transplantation
- Aggressive disease progression
Each case is evaluated individually based on clinical factors.
Effectiveness of CAR-T Therapy
CAR-T therapy is considered one of the most promising treatments for patients with limited therapeutic options. Clinical studies have demonstrated significant response rates, including complete remission in certain patients.
Response rates may vary depending on:
- Disease type
- Stage of disease
- Prior treatments
- Patient condition
- Molecular characteristics
Treatment decisions are made individually.
Possible Side Effects
CAR-T therapy may cause immune-related side effects. Most occur within the first weeks after infusion.
Common side effects include:
- Cytokine release syndrome
- Neurological symptoms
- Low blood counts
- Infection risk
- Fatigue
Most side effects are manageable with proper monitoring.
Limitations of CAR-T Therapy
Despite promising outcomes, CAR-T therapy has limitations:
- Patient eligibility criteria
- Complex preparation process
- Need for specialized centers
- Limited availability
Treatment decisions are individualized.
How Treatment Decisions Are Made
CAR-T therapy is considered after a comprehensive evaluation including:
- Disease type
- Disease stage
- Prior treatments
- Patient condition
- Laboratory findings
Decisions are typically made by a multidisciplinary team.
When to Consider Consultation
If you are considering CAR-T therapy, a specialist consultation can help evaluate treatment options and determine eligibility.
Contact for Consultation
Phone: +972-73-374-6844
WhatsApp: +972-52-337-3108
Email: [email protected]
Frequently Asked Questions
1. When is CAR-T therapy recommended?
In my practice, CAR-T therapy is usually considered when standard treatments are no longer effective. Most often, we evaluate this option in patients with relapsed disease or when the cancer does not respond to chemotherapy, targeted therapy, or even bone marrow transplantation.
I also consider CAR-T therapy for patients who have already undergone multiple lines of treatment and have limited remaining options. In these situations, CAR-T therapy may offer a meaningful alternative, particularly for certain types of lymphoma, leukemia, and multiple myeloma.
However, not every patient is a candidate. Before recommending CAR-T therapy, I carefully review the patient’s medical history, previous treatments, overall condition, and laboratory results. The final decision is always made individually after a comprehensive evaluation.
2. How long does CAR-T treatment take?
The full CAR-T therapy process typically takes 4 to 8 weeks, including:
- Patient evaluation
- Cell collection
- Manufacturing
- Chemotherapy preparation
- Cell infusion
- Monitoring period
3. Is hospitalization required?
Yes. Patients typically:
- Receive preparatory chemotherapy
- Stay in hospital 7–14 days
- Remain near the medical center for 2–4 weeks
4. Can the disease return?
Yes, relapse is still possible after CAR-T therapy. While many patients achieve remission, the long-term outcome varies depending on the type of disease, prior treatments, and individual response.
In my experience, some patients achieve durable remission that lasts for years, while others may require additional treatment later. This is why close follow-up after CAR-T therapy is essential.
After treatment, I usually recommend regular monitoring, including blood tests and imaging when necessary. If there are signs of disease progression, we evaluate additional treatment options and determine the next steps.
5. Can CAR-T be used after bone marrow transplant?
Yes, in certain cases CAR-T therapy may be considered after bone marrow transplantation. I typically evaluate this option when the disease relapses following transplant or when additional treatment options are limited.
Each situation is different. Before recommending CAR-T therapy after transplantation, I review factors such as the timing of relapse, previous treatments, patient condition, and potential risks.
For some patients, CAR-T therapy can be an important treatment option even after transplant. However, the decision is always individualized and based on a detailed medical assessment.